Atomic charges
A SODIUM ATOM WHICH CONTAINS ONE ELECTRON IN ITS VALENCE SHELL - IT’S UNSTABLE!
Remember that all atoms are striving to become stable! They become stable by having their valence shell full or empty of electrons.
Why do we call the elements neutral?
Thus far we have discussed atoms in the periodic table (aka elements) which are in their neutral form. The atoms contain the same amount of electrons as protons. They have not lost or gained any electrons. But, they are unstable!
Atoms want to be stable, to achieve this they will need to either gain or lose electrons in their valence shell, only then will atoms be considered stable.
When atoms lose or gain electrons it affects their net charge.
Atomic net charge:
The atomic net charge is equal to the proton number (+) added to the electron number (-) of an atom. The overall charge of an atom can be either negative, positive, or neutral (aka no charge).
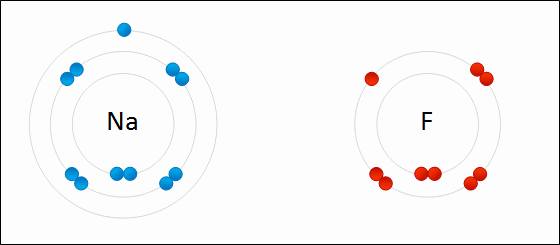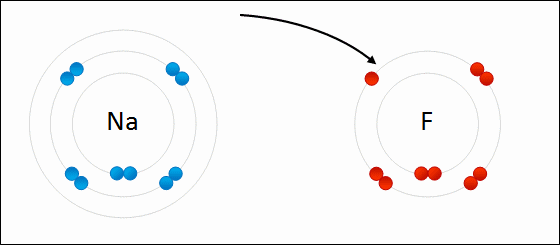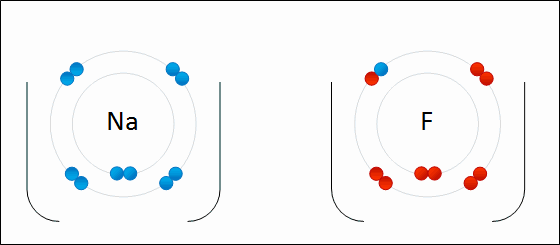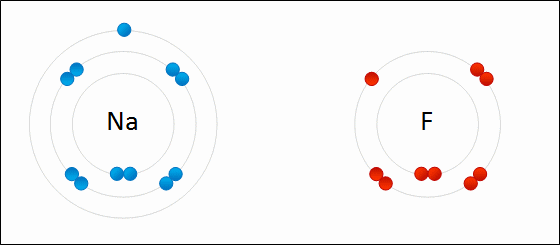
Ex. A sodium atom that has lost an electrons is (Na+)
Ex. A chlorine atom that has gained an electron is (Cl-)
What is the net charge of Sulfur that has gained 2 electrons? What is the net charge of Scandium that has lost 3 electrons?
Ions
Thus far we have been dealing with atoms which have the same number of electrons as protons.
To become more stable, atoms will try to empty or fill their farthest orbital (aka valence shell) with electrons. When atoms lose or gain electrons we call them ions.
ION:
Atoms that acquire an electrical charge through the loss or gain of electrons
If the atom loses electrons then it will acquire a positive charge (+)
If the atom gains electrons then it will acquire a negative charge (-)
There are special names for positive and negative ions:
Cation:
Atoms that lose electrons transform into positive ions (ex. Fe 2+, iron atom that has lost 2 electrons). Cations are attracted to negatively charged ions.
Anion:
Atoms that gain electrons transform into negative ions (ex. Cl 1-, chlorine atom that has gained 1 electron). Anions are attracted to positively charged ions.
To indicate an atom is an ion we place the charged number in superscript:
A calcium atom that lost 2 electrons would be Ca²⁺ (also know as a calcium cation)
A scandium atom that lost 3 electrons would be Sc³⁺ (also know as a scandium cation)
A sulfur atom that gained 2 electrons would be S²⁻ (also know as a sulfur anion)
A nitrogen atom that has neither gained nor lost electrons would be N⁰ - atoms that have a zero net charge are neutral, we do not indicate neutral/zero charges so in this case nitrogen would be indicated as plain old N.
If an atom has no charge is it called an ion?
Ionic Charge Patterns:
Alkali metals (group 1) have 1+ charge
Alkaline Earth metals (group 2) have a 2+ charge
Group 16 have mostly a 2- charge
Halogens (group 17) have a 1- charge
Noble gases (group 18) have no charge
Special Kinds Ions
Thus far we have been discussing single individual ions such as Be²⁺, the fancy scientific name for individual ions is monatomic ion.
Monatomic ion:
An individual atom that has a positive or negative charge (ex. Ba+ or I-).
There are a few exceptions though! When atoms buddy up and act as one single unit, such as (PO₄), we use the fancy scientific name of polyatomic ion.
Polyatomic ion:
A group of atoms that behave as a single unit with a positive or negative electrostatic charge, some examples include:
(PO₄)³⁻
(SO₃)²⁻
(H₃O)⁺
There is a list of polyatomic ions on the back of your Periodic Table of Elements sheet! Don’t let the complexity scare you!
Compounds
Where do atoms lose or gain electrons from? The answer is other atoms! Atoms are constantly swapping electrons to become more stable.
When atoms ‘trade’ or ‘share’ electrons they essentially bond to each other, this ‘buddying up’ is how compounds are formed.
Compound:
When two or more elements chemically combine (aka atoms share or transfer electrons) they form a compound.
When atoms ‘trade’ electrons they form an ionic bond, we refer to the ‘buddy’ grouping as an ionic compound. (ex. NaCl)
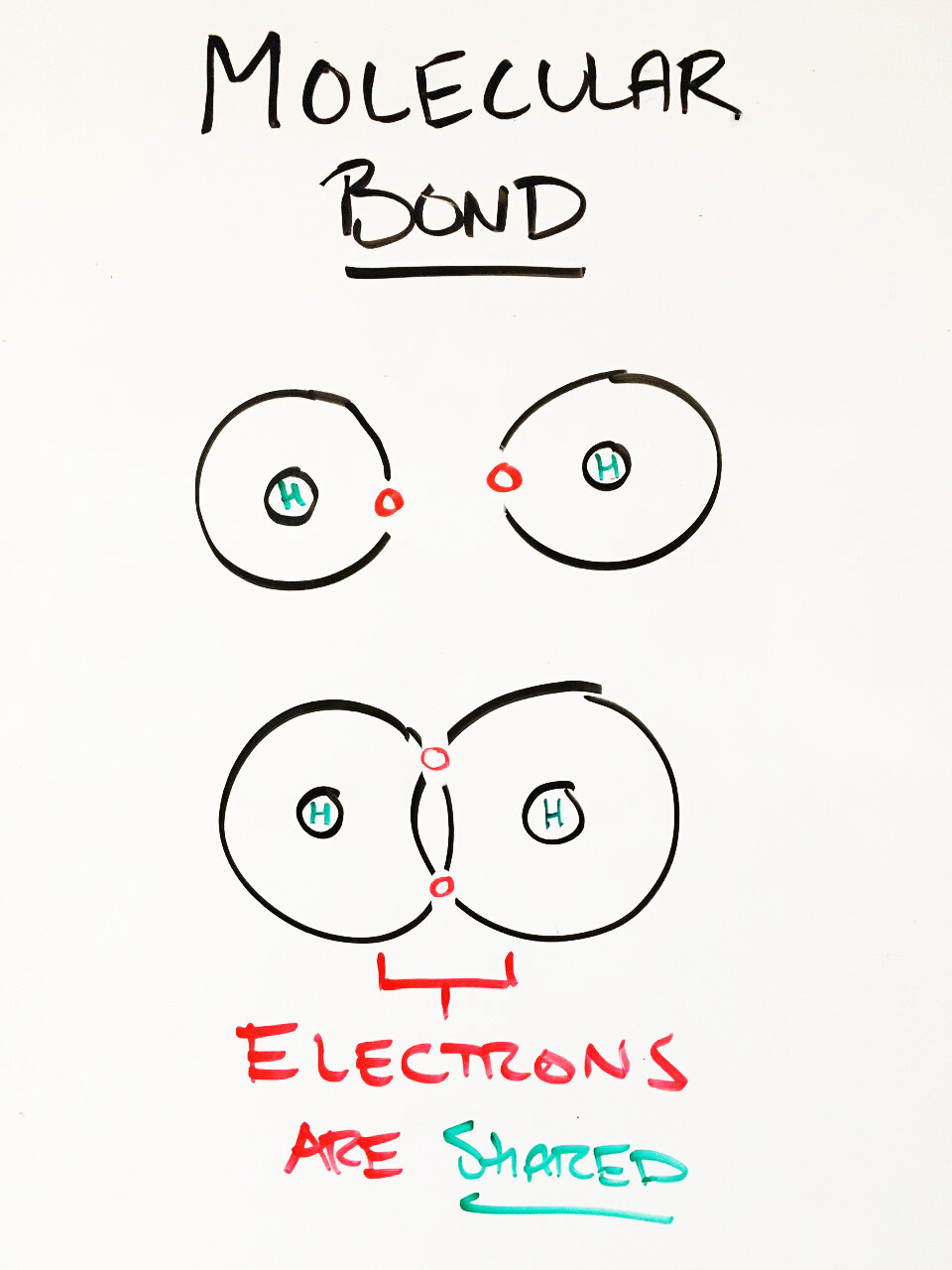
When atoms ‘share’ electrons they form a covalent bond, we refer to the ‘buddy’ grouping as a molecular compound. (ex. H₂O)
Ionic compounds
Remember when atoms interact with other atoms they may exchange electrons to become stable, and ‘buddy up’ to form compounds. Ionic compounds are formed when electrons are traded.
HOW A LITHIUM ATOM BONDS WITH A FLUORINE ATOM TO FORM LITHIUM FLUORIDE.
Ionic compound:
When two or more atoms transfer electrons they bond together. This bond creates an ionic compound and forms as a result of an attraction between particles of opposite charges (just like a magnet).
Ionic bonds involve cations (metals) donating/losing electrons to anions (non-metals).
Metals love donating electrons rather than sharing them.
Forming ionic compounds
Metal atoms bond to non-metal atoms!
Remember that ionic compounds are formed by one type of atom donating/losing an electron to another atom. The charge on elements considered to be metals are always positive (+) while the charge on non-metals are mostly negative (-). Thus metals love bonding with non-metals.
FORMATION OF SODIUM FLORIDE (NaF)
Some example of ionic compounds include:
Na⁺ bonds to F⁻ to form NaF (added to toothpaste to reduce cavities)
K⁺ and Br⁻ to form KBr (antisiezure drug)
Na⁺ and Cl⁻ to form NaCl (table salt)
Fe²⁺ and O²⁻ to form FeO (rust)
Characteristics of Ionic Compounds
Characteristics that all ionic compounds have in common are:
Ionic compounds are commonly known as ‘salts’ (this is different from table salt)
Ionic compounds form hard and brittle substances
Ionic compounds formed are solid at room temperature
Ionic compounds have high melting point and boiling point
Ionic compounds are good electrical conductor in an aqueous solution (dissolved in water)
Chemical formula terminology
Through hundreds of years compounds have been studied and chemicals have been named according to the scientist. Until the 18th century, no standardized system existed for naming chemicals.
In the 1780’s Guyton de Morveau began using the chemical symbol for each element to write out compounds. He decided the metal element in the compound is named first!
In 1920 IUPAC (International Union of Pure & Applied Chemistry) created the terminology for naming compounds and is now responsible for naming all chemical compounds discovered.
How to understand Chemical formulas
Three components of a chemical formula include:
1. Chemical symbols - identifies what element(s) are present in the compound
Ex. NaCl: one atom of sodium and one atom of chlorine
2. Subscript Numbers - indicates the number of atoms of elements that must combine to form the compound. It is placed below and after the symbol.
H₂O - 2 hydrogen, 1 oxygen
C₆H₁₂O₆ - 6 carbon, 12 hydrogen, 6 oxygen
3. Indicating Physical State of Element or Compound. After the chemical formula, as subscripts put: (s) for a solid compound, (l) for a liquid compound, (g) for a gaseous compound, (aq) compound dissolved in water
C₆H₁₂O₆ ₍s₎ or H₂O₍ʟ₎
Writing Ionic Compounds
SALT!
Naming Ionic Compounds
1. The chemical name of the metal (cation) comes first followed by the name of the non-metal (anion), the charges of each element are not written.
Ex. CaO is calcium oxide
2. Name of the non-metal (anion) changes to an ‘ide’ suffix.
Ex. Al2O3 - aluminum oxide
Ex. TiS₂ - titanium sulfide
* An exception to the ‘ide’ rule is if the anion is polyatomic, then the name of the polyatomic ion is used
Ex. CaSO₄ - calcium sulfate
3. If an element has multiple charges (ex. copper has a 1+ or 2+ charge), a Roman Numeral is added after the cation (Metal).
Ex. vanadium (IV) oxide
Ex. Iron (III) nitride
Ex. copper (II) Sulfate is CuSO4
Forming ionic compounds
ALWAYS have a copy of the Periodic Table
1. Print the metal element’s symbol with its ion charge, next to it, print the non-metal element’s symbol with its ion charge.
Ex: Ca2+ Cl1-
2. Balance the ion charges, the positive ion charge must balance the negative ion charges
Ex: Ca2+ Cl1- Cl1-
In the example, this means that there must be two chlorine atoms each with an ion charge of 1- to balance the 2+ ion charge of one calcium atom.
3. Write the formula by indicating how many atoms of each element are in it.
Do not include the ion charge in the formula!
Place the number of atoms of each element in a subscript after the element’s symbol.
If there is only one atom, no number is used
Ex:CaCl2
The Criss Cross Method!
Molecular Compounds
Thus far we have discussed how atoms can ‘trade’ electrons to become stable and form compounds. What if instead of trading they ‘shared’ electrons.
With molecular compounds electrons are shared. This means that electrons are neither lost nor gained by atoms. This also means atoms do not become ions! What elements share electrons? The non-metals are all about sharing!
Molecular compound:
When one or more non-metallic atoms combine they form a molecular compound (ex. CO, CO₂, H₂O). Another name for molecular compound is molecule.
Rather than donating electrons (like ionic compounds) molecular electrons share electrons, by sharing electrons atoms form a covalent bond.
There are no cations and anions when we discuss molecular compounds!
Characteristics of molecular Compounds
Molecular compounds characteristics differ greatly from ionic compounds characteristics:
WATER IS A MOLECULAR COMPOUND
Can be solids, liquids, or gases at room temp.
Poor electrical conductors
Low melting and boiling points
All molecular elements are non metallic
Diatoms
THE OXYGEN WE BREATHE IS DIATOMIC
Thus far we have discussed polyatomic ions (more than one atom), and monatomic (only one atom).
Remember non-metals love, love, love to share electrons. In fact many non-metals ‘buddy up’ with each other to form a somewhat stable pair. These molecules are considered to be diatoms.
Diatomic elements:
Elements that form molecules consisting of two atoms bonded together.
All halogens are diatomic, as well as N, H, and O:
Ex. Oxygen - O₂
Ex. Hydrogen - H₂
Ex. Chlorine - Cl₂
Molecules that are made of the same atom (ex. O₂) are homonuclear, while molecules made of different atoms are consider to be heteronuclear (ex. H₂O).
Naming Molecular Compounds
Similar to writing ionic compounds except:
No ions are present!
Ion charge not used in the formulas
Formula will show: What elements are present & proportion of atoms in a molecule
Ex. O₂₍g₎ has 2 oxygen atoms
IUPAC recommends that molecular compounds should be named using the prefix system only.
In the prefix system, Greek or Roman prefixes are used to indicate the number of each kind of atom bonded to one another
Molecular rules:
The first element in the formula should be named in full.
The second element in the formula should be shortened and given an ‘ide’ suffix…just like ionic compounds!
When there is more than one atom in the formula, a prefix is used which specifies the number of atoms.
The prefix ‘mono’ may be omitted on the first element
Forming Molecular Compounds
Predicting how molecular compounds will form is a much more complex process than with predicting ionic compounds, take for instance sugar C₁₂H₂₂O₁₁. How non-metals combine to form molecular compounds can be tricky to understand and is out of scope for grade 9 science. It’s best to save this lovely topic of molecular compounds for high school chemistry ;)
Chemical Reactions
KABLAMO! POTASSIUM NITRATE, CARBON, AND SULFUR COMBINE TO FORM GUN POWDER - AN INGREDIENT OF FIREWORKS
Recall The Evidence of a Chemical Reaction:
Color change
Odour
Gas or solid formation
Release or absorption of energy
Chemical Reactions :
When two or more ionic or molecular substances combine to form new substances
Atoms are rearranged
New bonds are formed
New substances with new properties form
Exothermic Reactions:
Chemical reactions that release heat.
Ex. Burning wood releases energy in the form of heat and light
Endothermic Reactions:
Chemical reactions that absorb energy. reactions need a continuous supply of energy.
Ex. A cold pack is triggered by a chemical reaction that absorbs heat from its surrounding.
We can express chemical reactions in 3 different ways…
Naming the chemical reaction: combustion of coal
Using a word equation: carbon + oxygen → carbon dioxide
Using a formula equation: C + O₂ → CO₂
Writing a formula Equation
Chemical reactions are often written as a formula equation. There are a few technical terms to know about formula equations:
Reactants: materials at the start of a reaction
Products: new materials produced by the reaction
An arrow indicates direction of reaction
A + B → C + D
(Reactants) → (Products)
A plus (+) sign is used to separate different reactants or products
Lets use the chemical equation for photosynthesis to illuminate the terms of a chemical equation
carbon dioxide + water → glucose + oxygen
CO₂ + H₂O → C₆H₁₂O₆ + O₂
The names of the reactants are written to the left of the arrow. The names of the products are written on the right of the arrow. Compounds are separated by a plus (+) sign.
The arrow can be read as “yields”, “forms” or “reacts to produce”
The Three c’s of Oxygen
Because oxygen is readily available and a very good electron acceptor it tends to be used be a common element used in chemical reactions.
Combustion (burning) – oxygen reacts with a fuel to produce carbon dioxide and water
Fuel (ex. wood) + oxygen → carbon dioxide + water
Cellular respiration (burning of sugar) - occurs in aerobic organisms (consumers that needs oxygen)
Food (ex. glucose) + oxygen → carbon dioxide + water
Corrosion (rusting) - a slow chemical change involving oxygen with some metals
Metal (ex. iron) + oxygen → rusted iron
Conservation of Mass in Chemical Reactions
CHEMICAL REACTION IN AN OPEN SYSTEM - NO STOPPER ON THE FLASK!
Open system:
An experiment in which the exchange of matter as well as energy interacts with the surroundings
Closed system:
An experiment in which all reactants and all products of a chemical reaction are accounted for
Law of Conservation:
Matter cannot be destroyed but can change form in a ‘closed’ controlled laboratory system.
Reactants → Products
100 g → 100 g
Mg + S → MgS
24.3g + 32.1 g → 56.4 g
Matter is not created nor destroyed during a chemical rxn. However, this being said, some matter such as gases may and usually escape and not be accountably measured in an ‘open system’ reaction as performed in most school labs
Balancing Equations
All chemical reactions must be balanced—the number of atoms, moles, and ultimately the total mass must be conserved during a chemical process.
1. Count the number of atoms on both the reactants and product sides of the equation
2. Using coefficients, balance the number of atoms. Always leave diatoms such as hydrogen and oxygen last
3. Check your work by counting the number of atoms on each side of the equation
Speed of Chemical Reactions
Chemical reactions can occur at different rates, take for instance rusting of metal which takes days, versus a firework which reacts within a matter of seconds. Certain factors can affect the speed that a reactions occurs at.
Recall that chemical reactions are essentially atoms interacting with each other, anything that increases the amount of interaction between atoms will increase reactions rates.
Four Factors affecting rate of reaction:
1. The presence of catalysts
Catalysts:
Any substance that increases the rate of a reaction without itself being consumed.
catalysts speed up reaction
not used in the reaction itself
enzymes in the body are a great examples of catalysts
2. Concentration of reactants
The greater the concentration, the faster the reaction. Why? More atoms to react with each other
3. Temperature of the reactants
Higher the heat, faster the reaction. Why? Causes the atoms to move faster creating more collisions with each other
4. Surface area of reactants
Greater surface area (ex. Powder vs chunks) the more surface area will be available for the reaction. More surface area means that more area is available in relation to the volume for reaction.